|
2/10/2024 0 Comments Nitrogen atomic emission spectrum
Found that metastable state of nitrogen molecules were the main factors leading to a series of excited state nitrogen atoms and nitrogen ionization. Under DC discharge, the strongest energy band N2 (C3π(u)), the weak Gaydon's Green system N2 (H3 -Φ(u)-G3 Δ(g)) and the emission line of nitrogen atoms (4 p-4 p0) at 820 nm were observed. Nitrogen emission spectra was observed at room temperature and low DC voltage (less than 150 V), and the emission spectrometry was used to diagnose the active species of the process of nitrogen discharge. Because the flame’s temperature is greatest at its center, the concentration of analyte atoms in an excited state is greater at the flame’s center than at its outer edges.A set of direct current (DC) discharge device of N2 plasma was developed, carbon nanotubes (CNT) modified ITO electrode was used as anode, aluminum plate as cathode, with -80 μm separation between them. An additional chemical interference results from self-absorption. These interferences are minimized by adjusting the flame’s composition and adding protecting agents, releasing agents, or ionization suppressors. Instruments may contain as many as 48–60 detectors.įlame emission is subject to the same types of chemical interferences as atomic absorption. A simple design for a multichannel spectrometer couples a monochromator with multiple detectors that can be positioned in a semicircular array around the monochromator at positions corresponding to the wavelengths for the analytes (Figure 10.59).įigure 10.59 Schematic diagram of a multichannel atomic emission spectrometer for the simultaneous analysis of several elements. This sequential analysis allows for a sampling rate of 3–4 analytes per minute.Īnother approach to a multielemental analysis is to use a multichannel instrument that allows us to simultaneously monitor many analytes. If the instrument includes a scanning monochromator, we can program it to move rapidly to an analyte’s desired wavelength, pause to record its emission intensity, and then move to the next analyte’s wavelength. Source: modified from Xvlun ().Ītomic emission spectroscopy is ideally suited for multielemental analysis because all analytes in a sample are excited simultaneously. This is accomplished by the tangential flow of argon shown in the schematic diagram.įigure 10.58 Schematic diagram of an inductively coupled plasma torch. At these high temperatures the outer quartz tube must be thermally isolated from the plasma. The resulting collisions with the abundant unionized gas give rise to resistive heating, providing temperatures as high as 10 000 K at the base of the plasma, and between 60 K at a height of 15–20 mm above the coil, where emission is usually measured. 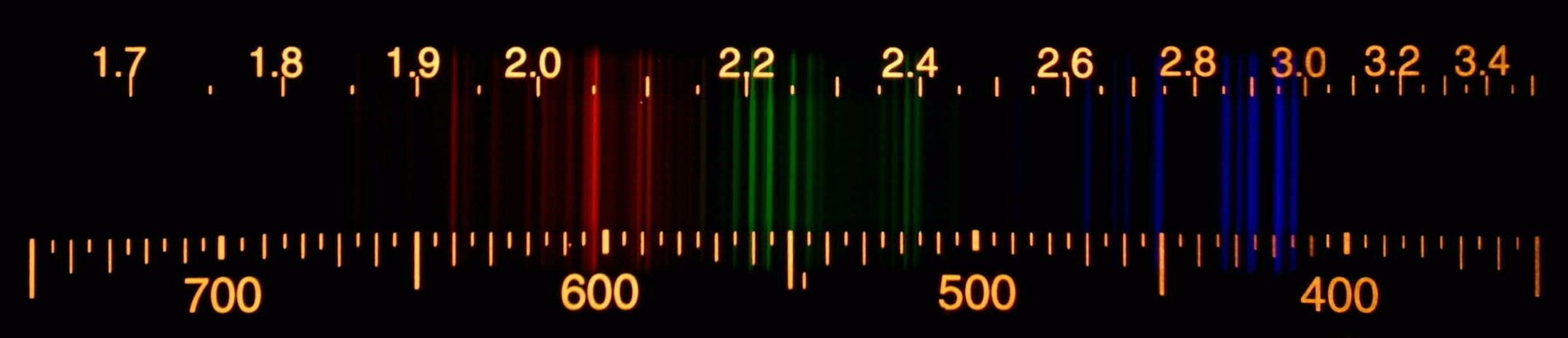
An alternating radio-frequency current in the induction coils creates a fluctuating magnetic field that induces the argon ions and the electrons to move in a circular path. Plasma formation is initiated by a spark from a Tesla coil. The sample is mixed with a stream of Ar using a nebulizer, and is carried to the plasma through the torch’s central capillary tube. The ICP torch consists of three concentric quartz tubes, surrounded at the top by a radio-frequency induction coil. Because plasmas operate at much higher temperatures than flames, they provide better atomization and a higher population of excited states.Ī schematic diagram of the inductively coupled plasma source (ICP) is shown in Figure 10.58. A plasma’s high temperature results from resistive heating as the electrons and argon ions move through the gas. The plasmas used in atomic emission are formed by ionizing a flowing stream of argon gas, producing argon ions and electrons. We also expect emission intensity to increase with temperature.Ī plasma is a hot, partially ionized gas that contains an abundant concentration of cations and electrons. From equation 10.31 we expect that excited states with lower energies have larger populations and more intense emission lines. Where g i and g 0 are statistical factors that account for the number of equivalent energy levels for the excited state and the ground state, E i is the energy of the excited state relative to a ground state energy, E 0, of 0, k is Boltzmann’s constant (1.3807 × 10 –23 J/K), and T is the temperature in kelvin.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
